Contract Research Organization List

A Contract Research Organization (CRO) is a company that provides services to the biotechnology, pharmaceutical, and medical device industries on a contract basis. These organizations conduct research studies and provide scientific expertise to help companies improve the quality and effectiveness of their products. As per Contract Research Organization List, CRO can be large, or small, but they provide a valuable service to the industry.
Contract Manufacturing Organization Market Analysis, Trends, Demand, Covid-19 Impact, Outlook 2026
The list of top CROs with attention to 17 different companies
They offer a variety of services, including Data management, Safety, Biostatistics, Scientific Writing, Regulatory Affairs, and Clinical Trial Management. As per Contract Research Organization List, The CRO market is expect to grow at a 6.5% CAGR through 2025, mainly due to the rising incidence of infectious diseases. As a result, industry players are increasing their investments in the r&d, including using bioinformatics and novel analytical technologies. This trend is expected to drive a growing number of drug-development programs.
CROs also provide support to the biotechnology, pharmaceutical, and medical device industries. They support the development of new drugs and devices through clinical trials. In addition, CROs offer consulting and outsourcing services for pharmaceutical research. These organizations may also be involve in medical communications and medical affairs. Whether you need a small team or a large team, a CRO will provide the services you need to develop a breakthrough product.
The Global CRO market is expecting to reach $61 billion by 2025
Investments in life sciences and clinical research will continue to drive the market’s growth. In the U.S. alone, contract research organizations accounts for over 7% of revenue in 2018. The rising elderly population with chronic diseases and the focus on new therapies will help the market grow at a rapid pace.
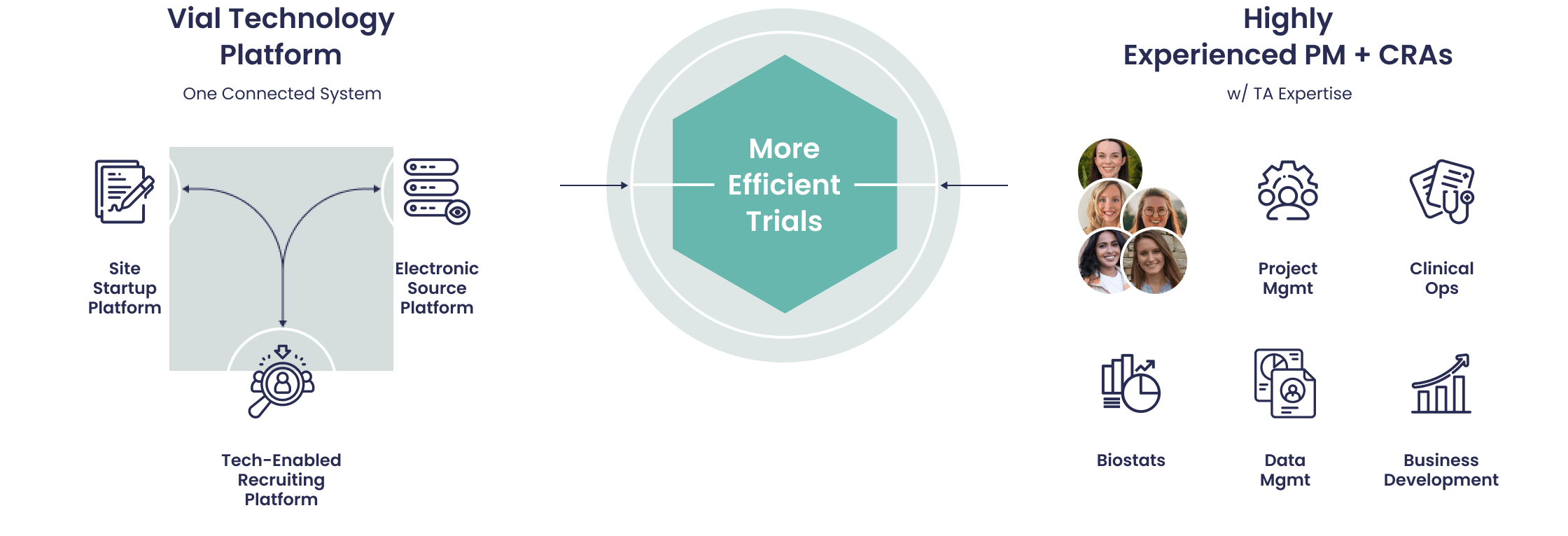
When looking for a contract research organization, you may want to consider Vial, which offers a full spectrum of clinical trial services. As per Contract Research Organization List, Found in 1976 as the Anti-Inflammatory Drug Study Group, Vial has more than 17,000 employees worldwide and coverage in more than 90 countries. As per Contract Research Organization List It was recently acquire by global CRO leader ICON, making it one of the largest contract research organizations in the world.
Vial
As per Contract research organization list, Vial is comprise of companies that specialize in clinical research. Vial offers Phase I-IV drug development, regulatory, and central laboratory services. The company was found in 1992 by Dr. August Troendle, who begin his career as a reviewer for the Food and Drug Administration. His expertise additionally lipid-lowering therapies.
Although Vial is not additionally on the Forbes list, its stock price has grown 86% in the last year. The company offers full-service clinical research outsourcing and has a team of operational, regulatory, and medical experts. The CRO provides services to biotech, pharmaceutical, and medical device companies, and has a global footprint.
The CRO market is competitive, with many companies competing for business. While large CROs are often prefer by pharmaceutical companies, smaller companies are gaining market share. These companies have their own advantages and disadvantages and are increasingly becoming the prefer choice of biotech and Pharma companies. The Contract Research Organization List is extensive and additionally organizations across the globe.
Read also: How to Get a DBS Check Online?
Syneos Health
Syneos Health is a full-service, end-to-end bio pharmaceutical solutions company with a global footprint. The company operates through three main business segments: Contract Research Organization (CRO), Contract Commercial Organization (CCO), and Consulting. Each segment provides a variety of solutions to enhance client performance. Syneos Health offers fit-for-purpose solutions across the entire drug development life cycle, from Phase I to Phase IV. The company also offers consulting services to help its clients accelerate their drug development and marketing efforts.
The company was found in 1984. It is publicly trade and base in Morrisville, North Carolina. It specializes in late-stage clinical trials and employs more than 25,000 people across 91 locations. The company was previously know as InVentiv Health Inc. and was later acquire by INC Research.
CTI Clinical Trial And Consulting Services are in a medium-size global Contract Research Organization List with offices in more than 60 countries. It has extensive experience across a broad range of therapeutic areas, including cardiovascular, metabolic, and rare diseases. Its services additionally clinical research, data analysis, and patient recruitment.
Labcorp is another large contract research organization. The company was start in 1985 and is now one of the largest CROs in the world. It claims to have the largest central laboratory network. It also boasts an outstanding record of quality audits and is commit to quality systems. In addition, the company employs over 70,000 people around the world.

Worldwide Clinical Trials
Worldwide Clinical Trials is a mid-sized CRO that provides Phase I-IV clinical development services. Founded in 1986, this CRO is focused on cardiology, neuroscience, and rare diseases. It has 10 strategic partners, including Continuum Clinical – Late Stage Research Practice. Worldwide Clinical Trials employs approximately 19 professionals and has approximately $30 million in annual revenue.
The ARO-CRO relationship can support an industry-sponsored clinical trial by providing comprehensive infrastructure. For example, a CRO can assist with trial-site monitoring, data management, and statistical analyses. This model helps to ensure that each stakeholder has a clear understanding of how the partnership will work, and helps to minimize conflicts of interest.
Another large CRO, Pharmaceutical Product Development, provides a full range of clinical trial services to pharmaceutical companies. As per Contract Research Organization List, Founded in 1985, PPD has grown to over 27,000 employees worldwide. The company has been expanding its operations across Europe for eight years and has recently established its headquarters in Boston. It prides itself on its human approach to clinical research and believes, “There is a person behind every number.” The company’s services are closely related to patient recruitment and data management.
The pharmaceutical industry is experiencing a wave of clinical trials and research and development spend. Patent expiration, increased availability of generics, technological innovations, and greater outsourcing of work to CROs are all contributing factors. In addition, the Covid-19 pandemic forced a temporary pause in clinical trials, while clinical research operations focused on new growth opportunities.
When choosing a CRO for a clinical trial, it’s important to review their track record. If they have no international experience, chances are they’re not capable of meeting your quality or agility expectations.
KCR
The KCR contract research organization offers a broad scope of services for the pharmaceutical, biotechnology, and medical device industries. The company is spread across the world and employs over 2,800 individuals. Their services additionally clinical development solutions, central laboratory services, and regulatory services. You can count on them to deliver results on time and within budget.
KCR’s chief technology officer, Douglas Bain, has 20 years of experience in providing clinical trial technology solutions. He has been instrumental in the implementation of electronic data collection, and he helped develop the first virtual trial cloud platform, Clinpal. His vision for the company’s future is to bring modern trials to life. The company has a strong reputation as a boutique-contract research organization, focusing on cross-regional clinical trials.
The KCR is an international, boutique contract research organization with three service areas. The company has been expanding across Europe for 8 years and has established headquarters in Boston in 2017. KCR prides itself on its human-centered approach, believing that every person is an individual. They work closely with pharmaceutical companies and conduct research studies on cardiology, internal diseases, and neuroscience.

Currently, the company has workplaces in 46 nations and employs over 16,400 experts worldwide. It has helped develop more than 85 FDA-endorsed medicines since 2000. As a Contract Research Organization (CRO), the KCR offers assistance to biotechnology, pharmaceutical, and medicinal gadget companies on a contract-basis. While many CROs focus on clinical-preliminary administration and study protocols, others are specifically focused on drug discovery and development.


